Category: Medical Devices
“Get This Thing Out of My Chest”
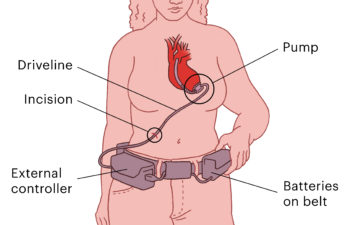
A life-sustaining heart pump was taken off the market after years of problems and FDA inaction. Thousands of people are now stuck with it embedded in their hearts.
Beware of Fraudulent Coronavirus Tests, Vaccines and Treatments
The FDA is particularly concerned that these deceptive and misleading products might cause people to delay or stop appropriate medical treatment for COVID-19, leading to serious and life-threatening harm
Thousands of Patients Were Implanted With Heart Pumps That the FDA Knew Could Be Dangerous
Inspectors repeatedly found manufacturing and device quality problems with the HeartWare pump. But the FDA didn’t penalize the company, and patients had the device implanted without knowing the facts.
Hidden FDA Reports Detail Harm Caused By Scores Of Medical Devices
FDA lets medical device companies file reports of injuries and malfunctions out of a widely scrutinized public database, leaving doctors in the dark.