Sarah L Caddy, University of Cambridge
Many options are being explored to treat COVID-19. These include new drugs specifically designed to target SARS-CoV-2, as well as “repurposed” drugs – that is, existing drugs designed to treat a different disease.
By far the oldest treatment being tested, though, is convalescent plasma. This involves using blood plasma from people who have recovered from COVID-19 and infusing it into patients who currently have the disease.
Plasma is the liquid portion of blood that remains when all red and white blood cells and platelets have been removed. It was over a hundred years ago that Emil Behring was awarded the first Nobel prize for physiology and medicine for his work demonstrating that plasma could be used to treat diphtheria.
We now know that the key component of plasma for treating infections is antibodies. Antibodies are Y-shaped proteins that are highly specific for whichever infection a person has previously encountered. They bind to proteins from the infectious agent, called antigens.
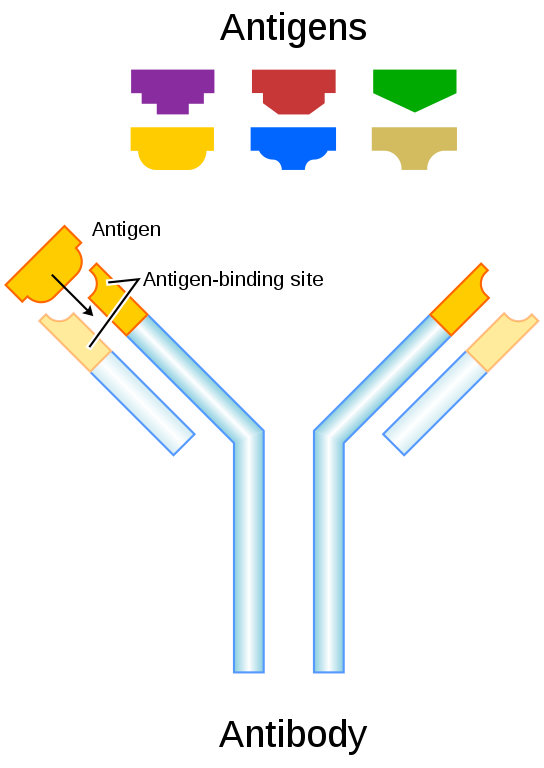
They are produced in vast quantities by B cells of our immune system in order to bind to the invading virus and then target it for destruction. The concept of vaccination relies on stimulating antibody production to infections not yet met.
In contrast, using convalescent plasma involves the transfer of antibodies from donors who have already mounted an immune response, thus offering immediate (but transient) protection to the recipient.

Wikimedia Commons
Convalescent plasma has been trialled as a therapy in previous coronavirus outbreaks. A few observational studies were conducted during the first Sars epidemic in 2003.
These all reported improvement in patients after receiving convalescent plasma, and no evidence of serious complications. However, these studies were largely case reports – not the most reliable type of evidence.
Convalescent plasma treatment was also tested during the Ebola virus outbreak in 2013-2016. Several case reports showed promising results, but again, large-scale randomised trials were not performed. Still, the World Health Organization published guidelines on the appropriate use of plasma from recovered patients.
The handful of early reports in which COVID-19 patients have been treated with convalescent plasma have garnered plenty of interest. Each has concluded that plasma therapy is safe and improves patient outcome, but there are significant limitations to each of these studies.
To begin with, each study has only treated a maximum of ten patients. Also, there were no control patients (people who weren’t given convalescent plasma), so it’s impossible to know how the patients may have responded without treatment.
Fortunately, more extensive studies are now in the pipeline to provide robust evidence either for or against the use of convalescent plasma. Worldwide there are over 60 clinical trials actively recruiting COVID-19 patients to study the effect of convalescent plasma.
Patients are typically given about 500ml plasma intravenously, and their progress is then carefully monitored. Many studies are using plasma from non-infected patients as the placebo arm of the trial, to ensure that any benefits identified are indeed specific for SARS-CoV-2 antibodies.
Although convalescent plasma has the potential to be a useful treatment, there are a few theoretical concerns that will keep all clinical trial teams on their toes.
First, artificially supplying antibodies might make an infection worse. This is due to a rare phenomenon called antibody-dependent enhancement (ADE). Antibodies that bind to a virus may be taken up by cells expressing antibody receptors.
This could enable the virus to enter cells that are not normally susceptible to infection, which may increase the number of new viral particles made. This classically occurs in dengue infection, but has also been a concern for MERS coronavirus.
A second theoretical risk is that pre-formed antibodies may block the body’s own immune response from responding adequately. We know this occurs with maternal antibodies; antibodies passed naturally from mother to offspring can prevent infants from responding correctly to vaccination. This is why most childhood vaccines are started after eight weeks of age.
How safe is convalescent plasma? It’s important to bear in mind that there are several potential risks with receiving plasma from another person. Mild side effects include developing a fever or allergic reactions, such as a rash and an itchy sensation. The plasma should also be compatible with the recipient’s blood group to prevent transfusion reactions.
Much more serious is the risk of transmitting blood-borne infections. Fortunately, the risk can be minimised by screening the donor for infections.
For example, in the UK, blood donation is centralised by the NHS, and all donors are rigorously screened for syphilis, HIV, hepatitis B, C, E, and HTLV-1 (human T cell leukaemia virus 1) to ensure the risk of transmitting these infections are minimal.
Another limitation to consider is that the elderly population with weakened heart or lungs (those at high risk from COVID-19), may not tolerate receiving such a large volume of plasma. This can lead to a complication known as “transfusion associated circulatory overload”.
Despite its 100-years-plus history, never before has the use of convalescent plasma been so thoroughly tested for use as a viable therapeutic. Whatever the outcome of the many clinical trials being conducted, we will certainly learn a lot about the most efficient ways to collect, process and safely use convalescent plasma.
Fingers crossed for a positive outcome for convalescent plasma use in COVID-19. Hopefully, our improved understanding of convalescent plasma will also be valuable for pandemics of the future.
Sarah L Caddy, Clinical Research Fellow in Viral Immunology and Veterinary Surgeon, University of Cambridge


