Category: Biotechnology
AI inconsistently assesses cardiac risk from chest pain – UW study finds
Tasked to interpret data associated with patient complaints of nontraumatic chest pain, the ChatGPT-4 large language model performed poorly against two standard tools that doctors use to predict risk of a cardiac event.
AI shown to dramatically speed protein engineering – UW study
Protein engineering scientists have been able to design more efficient proteins using machine learning quickly, dramatically shortening a process that typically takes months to years of trial and error.
First Cancer TIL Therapy Gets FDA Approval for Advanced Melanoma
In this treatment, immune cells from the patient’s tumor are collected and expanded in culture and then infused back into the patient.
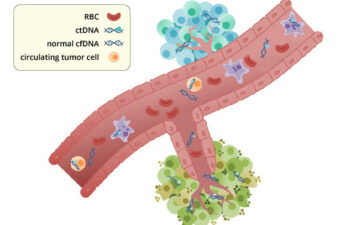
By looking for fragments of DNA that have escaped from tumors into the bloodstream, or circulating tumor DNA (ctDNA), doctors may be able to identify which of their patients with colorectal cancer need chemotherapy after surgery.
Several companies are testing brain implants – why is there so much attention swirling around Neuralink? Two professors unpack the ethical issues
Putting a computer inside someone’s brain used to feel like the edge of science fiction. Today, it’s a reality. Academic and commercial groups are testing “brain-computer interface” devices to enable people with disabilities to function more independently. Two two scholars at the University of Washington School of Medicine – Nancy Jecker, a bioethicst, and Andrew Ko, a neurosurgeon who implants brain chip devices – discuss the ethics of this new horizon in neuroscience.
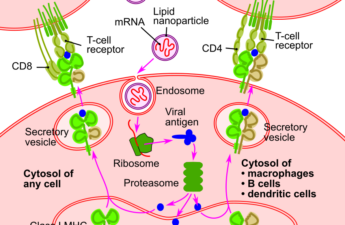
Nobel prize in medicine awarded to mRNA pioneers – here’s how their discovery was integral to COVID vaccine development
Dr Katalin Karikó and Dr Drew Weissmanhave been awarded the Nobel Prize for their discoveries into mRNA biology. The pair were the first to discover a way of modifying mRNA that allowed it to successfully be delivered to cells and replicated by them. Their discovery was not only integral to COVID-19 vaccine development, but may also lead to the development of many other therapies – such as vaccines for cancer.
Monitoring brainwaves to track how patients with depression respond the treatment
Depression recovery can be hard to measure − new research on deep brain stimulation shows how objective biomarkers could help make treatment more precise Christopher Rozell, Georgia Institute of Technology and Sankaraleengam Alagapan, Georgia Institute of Technology It can be challenging…
Experts Debate the Risks of Made-to-Order DNA
By one expert’s estimate, perhaps 30,000 scientists worldwide have the skills to build a strain of pandemic influenza, provided they can find someone to synthesize the DNA for them. The consequences of unleashing such a pathogen could be catastrophic.
The ethics of brain-computer interfaces lag behind the science, write a philosopher and a neurosurgeon from UW.
Researchers are exploring nonmedical brain-computer interface applications in many fields, including gaming, virtual reality, artistic performance, warfare and air traffic control.
A blood test that screens for multiple cancers at once promises to boost early detection
Testing to look for circulating tumor DNA in the blood is not new. These liquid biopsies – a fancy way of saying blood tests – are already widely used for patients with advanced-stage cancer. Doctors use these blood tests to look for mutations in the tumor DNA that help guide treatment.
Newly available over-the-counter hearing aids offer many benefits, but consumers should be aware of the potential drawbacks
It seems the over-the-counter hearing aids would be a great solution for patients with hearing loss, right? Less hassle and less cost – in many cases, thousands of dollars less – and more people than ever getting the help they need. But it’s not that simple.
Say What? Hearing Aids Available Over-the-Counter for as Low as $199, and Without a Prescription
Starting Monday, consumers will be able to buy hearing aids directly off store shelves and at dramatically lower prices as a 2017 federal law finally takes effect.
NIH initiative to systematically investigate and establish function of every human gene
Creating a catalog of what all human genes do is no easy feat. Most genes are likely to have more than one function and behave differently depending on the type of cell in which they are expressed. In addition, genes may turn on or off depending on the cell’s relationship to surrounding cells, environment and age.
Using nanotechnology and AI to diagnose TB in children
Combining nanotechnology with artificial intelligence can diagnose tuberculosis in children in whom the deadly disease might otherwise go undetected.
Scientists set out to map the world’s genomic diversity
“The goal is to collect, organize and make accessible a representation of all the genetic variation that exist in humans, big, small, common and rare,” said Evan Eichler, a professor of genome sciences at the University of Washington School of Medicine in Seattle and one of the organizers of the project.