Category: Genetics
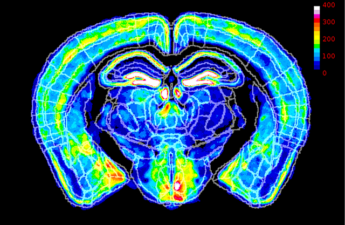
X-chromosome inactivation may reduce autism risk, UW study finds
In mice, the pattern of X-chromosome inactivation in female brains altered the effect of a mutation linked to the disorder.
Your biological age predicts dementia and stroke regardless of your actual age – new study
while we all grow older chronologically at the same pace, biologically, our clocks can tick faster or slower. Relying solely on chronological age – the number of years since birth – is inadequate to measure the body’s internal biological age.
COVID origins debate: what to make of new findings linking the virus to raccoon dogs
While this latest data is one additional piece of the puzzle that supports an origin of the pandemic linked to Wuhan’s animal trade, it is unlikely to provide irrefutable evidence.
Epigenetic and social factors both predict aging and health – but new research suggests one might be stronger
For years, researchers have been using clinical factors normally collected at physicals, like hypertension, cholesterol and weight, as indicators to predict aging. The idea was that these measures could determine whether someone is a fast or slow ager at any point in their life cycle. But more recently, researchers have theorized that there are other biological markers that reflect aging at the molecular and cellular level. This includes modifications to a person’s genetic material itself, or epigenetics.
Genetics might explain why some people have never had COVID
. . . but we shouldn’t be too focused on finding out
Experts Debate the Risks of Made-to-Order DNA
By one expert’s estimate, perhaps 30,000 scientists worldwide have the skills to build a strain of pandemic influenza, provided they can find someone to synthesize the DNA for them. The consequences of unleashing such a pathogen could be catastrophic.
A blood test that screens for multiple cancers at once promises to boost early detection
Testing to look for circulating tumor DNA in the blood is not new. These liquid biopsies – a fancy way of saying blood tests – are already widely used for patients with advanced-stage cancer. Doctors use these blood tests to look for mutations in the tumor DNA that help guide treatment.
NIH initiative to systematically investigate and establish function of every human gene
Creating a catalog of what all human genes do is no easy feat. Most genes are likely to have more than one function and behave differently depending on the type of cell in which they are expressed. In addition, genes may turn on or off depending on the cell’s relationship to surrounding cells, environment and age.
Scientists set out to map the world’s genomic diversity
“The goal is to collect, organize and make accessible a representation of all the genetic variation that exist in humans, big, small, common and rare,” said Evan Eichler, a professor of genome sciences at the University of Washington School of Medicine in Seattle and one of the organizers of the project.
When it comes to the rarest of diseases, the diagnosis isn’t the answer – it’s just the starting point
Major advances in the precision and speed of gene sequencing technology followed by dramatic reductions in the costs of testing – have radically changed how medical genetics clinics function. But while sequencing can provide confirmation of a suspected, well-understood condition, it frequently results in a situation like that faced by the Smiths, where the testing result shows an incredibly rare disorder with little known about it.
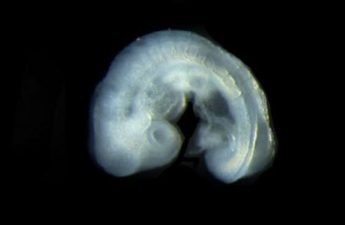
UW scientists create ‘roadmap’ tracing the course of embryo cell development
This “roadmap” of mouse embryogenesis will help researchers understand the molecular programs that control how different specialized cell types emerge from less differentiated cells as an embryo grows and develops.
5 things research from twins taught us about health, behaviour and what makes us unique
The first major insight is that genes and environments almost always combine to influence our life trajectory. Sometimes the largest factor is genetics (think genetic disorders). Sometimes it’s environment (think infections). Mostly, it’s somewhere in between.
Men should check their genes for prostate cancer risk, UW researchers say.
Approximately 1 in 10 men with advanced prostate cancer carries an inherited gene mutation, according to UW researchers.
UW scientists to study effects of variations in the human genome
Researchers seek to determine how variations in the DNA sequences in the human genome affect how cells’ function and, ultimately, how these variants influence human health and disease.