Category: Drugs
States struggle to help patients navigate insurance hurdle known as ‘step therapy’
Millions of Americans have experienced similar frustrations under protocols known as step therapy, or fail-first policies. Insurance companies, and the pharmacy benefit management companies that handle prescriptions for them, often refuse to cover a specific drug until after the patient has tried cheaper alternatives. Insurers argue that step therapy — taking drug treatment one step a time — prevents wasteful spending by directing patients to less expensive, but still effective, treatments.
Masks work, our comprehensive review has found
The more people wore their masks, the more effective the masks turned out to be.
Alcohol use disorder can be treated with an array of medications – but few people have heard of them
While 22% of patients with opioid use disorder receive medications to treat it, the rate of medication treatment for alcohol use disorder is much lower. Less than 10% of people with alcohol use disorder receive any treatment in any year, and less than 3% receive medications for it.
More than 321,000 U.S. children lost a parent to drug overdose from 2011 to 2021
The highest number of affected children were those with non-Hispanic white parents, but communities of color and tribal communities were disproportionately affected.
Need a rapid health test? Try our kiosks.
Washington State Department of Health (DOH)-sponsored kiosks with free COVID-19 tests, flu tests, and more are popping up all over Washington. Let’s dig into the what, where, and why.
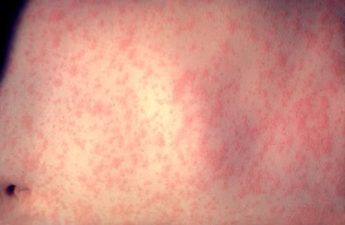
PUBLIC HEALTH MONITORING MEASLES EXPOSURE AMONG INTERNATIONAL TRAVELERS VISITING SEATTLE
Last week that a group of international travelers visiting Seattle were exposed to an individual with measles prior to arriving in Seattle. Currently, there are no cases of measles among the group (or within King County), but Public Health — Seattle & King County is monitoring the situation closely.
Psychedelics could make mental health worse in people with a personality disorder
Various personality disorders might respond differently to psychedelics. For instance, people with histrionic personality disorder (excessive attention-seeking and emotional overreaction) or borderline personality disorder (emotional instability, intense relationships and fear of abandonment) might feel worse or more unstable. And those with schizotypal personality disorder (social anxiety, odd beliefs and eccentric behaviour) could become more paranoid. People with narcissistic personality disorder (excessive self-importance, lack of empathy, and need for admiration) may struggle with the self-reflective nature of psychedelics because they often have a hard time handling criticism.
Surgery won’t fix my chronic back pain, so what will?
Treatments offered to people with chronic pain include strong pain medicines such as opioids and invasive procedures such as spinal cord stimulators or spinal fusion surgery. Unfortunately, these treatments have little if any benefit and are associated with a risk of significant harm.
Nitazenes found in 5 overdose deaths in Philly – here’s what they are and why they’re so deadly
Researchers have relatively little information on how the human body reacts to nitazenes because the drugs have never gone through clinical trials. But lab tests show certain nitazenes could be hundreds to thousands of times more potent than morphine and 10 to 40 times stronger than fentanyl.
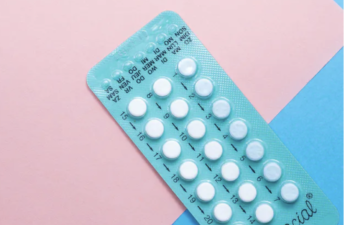
Social media will tell you birth control causes mental health issues, weight gain and infertility – here are the facts
In recent years, there’s been a rise in misinformation about hormonal contraceptives on social media. Some women are reportedly even stopping their birth control as a result of misleading posts they’ve seen on TikTok and Instagram.
Drugs like Ozempic won’t ‘cure’ obesity but they might make us more fat-phobic
Ironically, while fat people are told they need to lose weight for their health, they are also shamed for “cheating” or taking shortcuts by using medication.
Know Which Medication Is Right for Your Seasonal Allergies
An allergy is your body’s reaction to an otherwise innocent substance that it has identified as an invader. If you have allergies and encounter a trigger (allergen), your immune system fights it by releasing chemicals, such as histamine (hence the term “antihistamine”). Histamine causes symptoms, such as runny nose, itchy nose, sneezing, and itchy and watery eyes.
Considering taking a weight-loss drug like Ozempic? Here are some potential risks and benefits
As they’ve grown in popularity, we’ve also heard more about the potential side effects – from common gastrointestinal discomforts, to more serious mental health concerns.
King County launches ‘bup’ hotline.
Buprenorphine, also called suboxone, is a medication used to treat opioid use disorder. It is one of the best available treatments to alleviate withdrawal, reduce cravings, and reduce overdose risk by about half when taken as directed. It works by binding to the same receptors that opioids like fentanyl bind to, but it only turns them on about halfway. That keeps people from feeling sick and helps with their cravings.
Cardiovascular risks and COVID-19: New research confirms the benefits of vaccination
A new study found that common cardiovascular complications of COVID-19 were substantially reduced in people who were vaccinated, with protective effects lasting up to a year after vaccination.